What is subcutaneous pembrolizumab and what does the NHS rollout mean for people living with lung cancer in Europe?
What is subcutaneous pembrolizumab and what does the NHS rollout mean for people living with lung cancer in Europe?
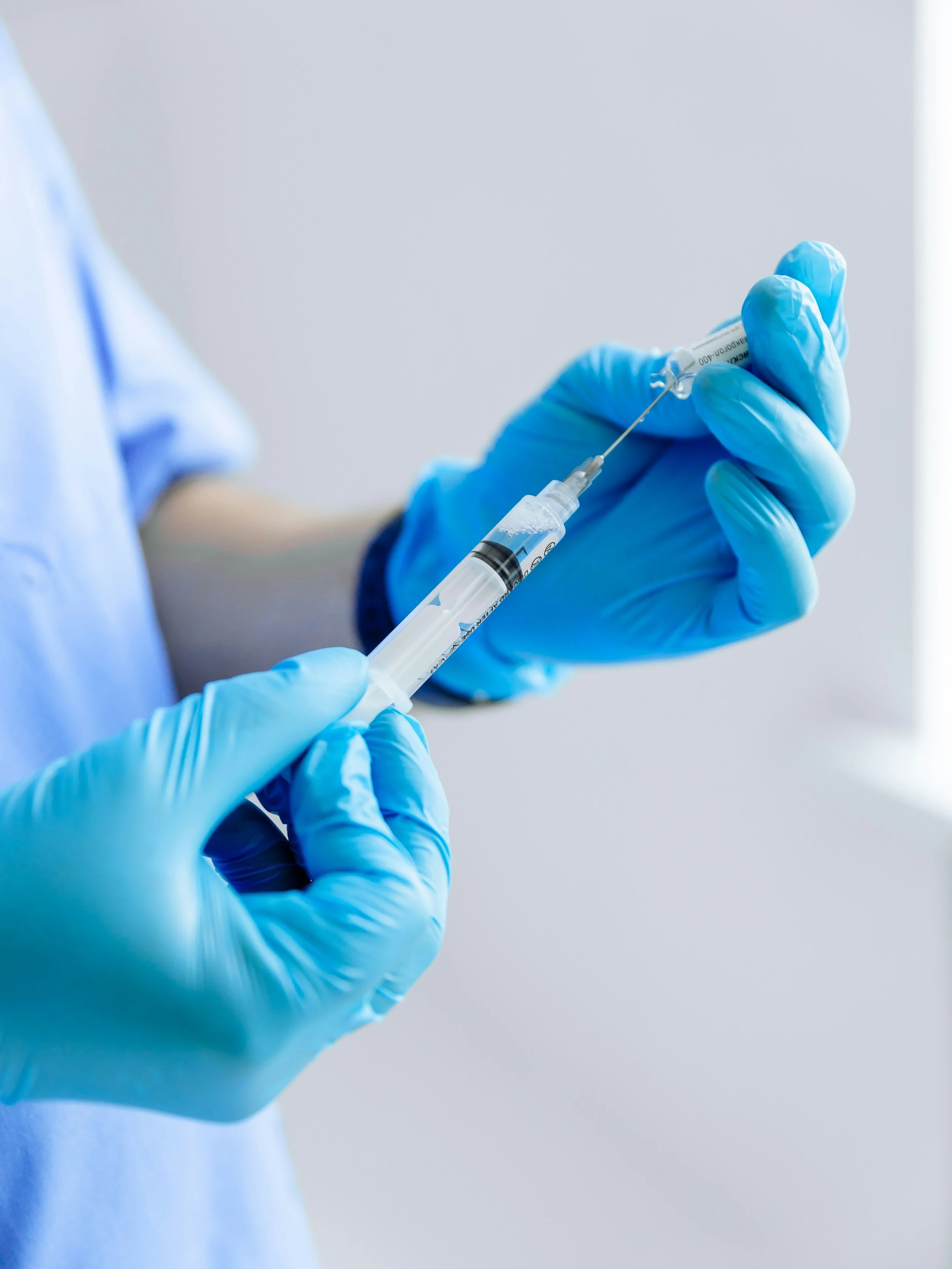
On 4 May 2026, NHS England began rolling out a new injectable form of pembrolizumab - one of the most widely used immunotherapy treatments for lung cancer. For people living with lung cancer who receive this treatment, it marks a significant practical change: a one-minute injection replacing an intravenous infusion that can take up to two hours. Treatment time is cut by up to 90%.
This is positive progress. It is also a prompt to ask a broader question: when will people living with lung cancer across Europe have the same access?
What is pembrolizumab and how is it used in lung cancer treatment?
Pembrolizumab, marketed as Keytruda, is a type of immunotherapy known as a PD-1 inhibitor. Cancer cells can evade the body’s immune system by producing proteins that signal immune cells not to attack. Pembrolizumab works by blocking that signal, allowing the immune system to recognise and destroy cancer cells.
In lung cancer treatment, pembrolizumab is one of the most important medicines available. It is used as a first-line treatment for people with non-small cell lung cancer (NSCLC), both as a monotherapy in people with high PD-L1 expression and in combination with chemotherapy. Around 14,000 people in England start a course of pembrolizumab each year. It is now the world’s best-selling prescription medicine, with global sales of $31.68 billion in 2025.
The subcutaneous formulation works in the same way as the intravenous version. Clinical trial data from the phase 3 3475A-D77 trial, conducted specifically in people with metastatic NSCLC, confirmed that it delivers equivalent drug exposure and comparable outcomes.
Why does reducing treatment time matter for people living with lung cancer?
For people managing lung cancer treatment alongside everyday life, the difference between a one-minute injection and two hours in a hospital chair is not a minor detail.
Treatment is exhausting. Regular hospital visits add to that burden - the travel, the waiting, the time away from work, family and the things that make life meaningful. Reducing the time spent on each treatment visit directly improves quality of life during what is already a demanding period.
Our own research underlines why this matters. The 11th Lung Cancer Europe Report found that 89% of people living with lung cancer experience emotional difficulties after diagnosis. Reducing treatment time is not a minor administrative improvement, it is a quality of life intervention.
At Lung Cancer Europe, we believe that living longer should also mean living better. Quality of life is not a secondary consideration in cancer care - it is a core part of it. The NHS rollout of subcutaneous pembrolizumab is a practical example of what it looks like when that principle is taken seriously.
There are also system benefits. The injectable form reduces pharmacy preparation time by around 44% and frees up capacity in chemotherapy units. NHS England estimates staff could save more than 100,000 hours a year. That matters for waiting times and for the sustainability of cancer services.
Is subcutaneous pembrolizumab approved in Europe?
Yes. The European Commission approved the subcutaneous formulation of pembrolizumab, marketed as Keytruda SC, in November 2025. The approval covers all 33 adult indications for which intravenous pembrolizumab is approved in Europe, and is valid across all 27 EU member states as well as Iceland, Liechtenstein and Norway.
The approval was supported by data from the phase 3 3475A-D77 trial, which was conducted in people with treatment-naive metastatic NSCLC and confirmed that the subcutaneous formulation delivers non-inferior pharmacokinetics compared to the intravenous form.
In short: the science is established, and the regulatory decision has been made.
When will people living with lung cancer in Europe be able to access it?
This is where the picture becomes less clear.
European Commission approval authorises the marketing of a medicine across member states. It does not guarantee that people can access it. Commercial availability in each country depends on national reimbursement procedures, which vary significantly across Europe and are still ongoing in most countries.
This is a pattern that Lung Cancer Europe knows well. In some European countries, people can wait more than 600 days for access to newly approved medicines. The gap between regulatory approval and real-world access is one of the most persistent inequalities in cancer care across Europe.
The NHS rollout today demonstrates what is possible when a health system moves quickly. Subcutaneous pembrolizumab was approved in Europe in November 2025. People living with lung cancer across Europe should not have to wait significantly longer to benefit from the same advance.
Breakthroughs only matter if people can benefit from them. Access to treatment should be equitable across Europe, regardless of geography or ability to pay.