EMA recommends tarlatamab for relapsed extensive-stage SCLC
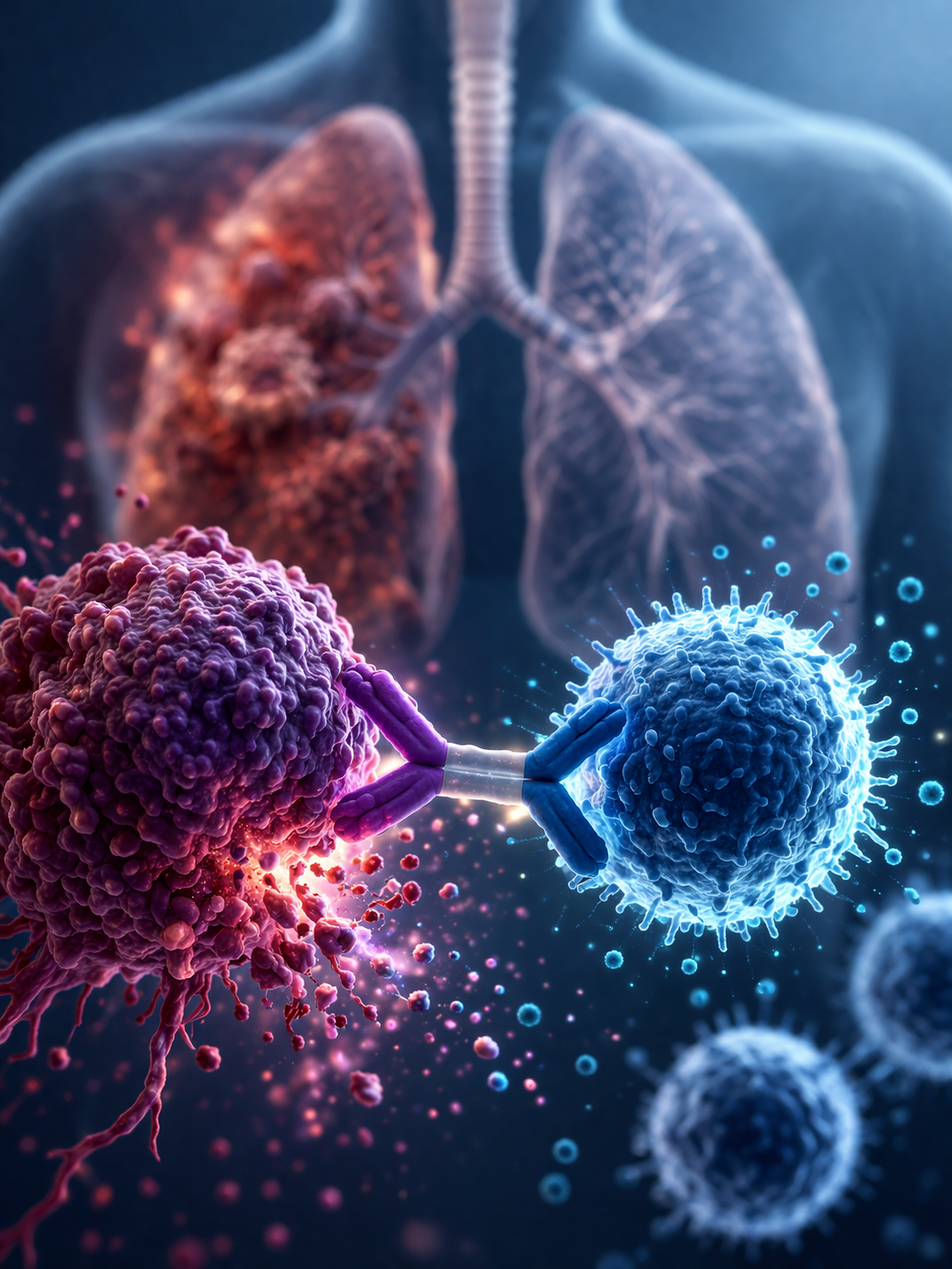
The European Medicines Agency has recommended a marketing authorisation for Imdylltra (tarlatamab) for adults with extensive-stage small cell lung cancer whose disease has relapsed during or after first-line platinum-based chemotherapy. EMA says this follows phase 3 data showing improved overall survival compared with standard-of-care chemotherapy in this setting.
In the study cited by EMA, median overall survival was 13.6 months with tarlatamab compared with 8.3 months with standard of care. Median progression-free survival was 4.2 months versus 3.2 months. Tarlatamab is a bispecific antibody that targets DLL3 on tumour cells and CD3 on T cells, helping bring the immune system into direct contact with the cancer.
This is an important regulatory step for small cell lung cancer, where treatment options after relapse remain limited. The CHMP opinion now goes to the European Commission for a final decision on EU-wide marketing authorisation. After that, access will still depend on national pricing and reimbursement decisions in each country.